How To Draw Newman Projections From Bond Line
Bail Line Notation to Newman Project
Key Questions
-
The bond line structure of ethane is

The wedge-dash structures for the staggered and eclipsed conformations are:
Here'due south how to convert the wedge-dash structures to Newman Projections.
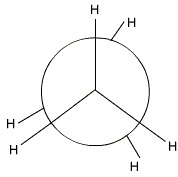
Staggered Ethane
Step ane. View the molecule along the C1-C2 axis with your eye at the left-mitt end.
The closest bonds make an inverted Y, so you will choose that template for your project.
Step 2. Place the groups onto your template.
All groups on the carbon atoms are H atoms.
And you accept your staggered Newman projection for ethane.
Eclipsed Ethane
Step 1. Describe two eclipsed Newman templates.
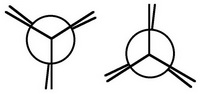
Step 2. View the molecule along the C1-C2 axis with your eye at the left-hand end.
The closest bonds make an inverted Y, then you will cull the 2nd template for your project.
Footstep 3. Place the groups onto your template.
All groups on the carbon atoms are H atoms.
And y'all have your eclipsed Newman projection for ethane.
-
A bond-line notation gives no stereochemical information, merely here'southward how to catechumen a wedge-dash notation to a Newman projection.
Problem
Convert the following wedge-dash structure of 2-chloro-three-methylpentane to a Newman project through the C2-C3 bond.
Pace ane. Draw two Newman projection templates.
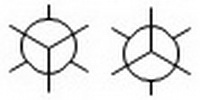
- Draw two circles.
- Put a dot in the centre of each. These correspond C-2. C-three is hiding backside the circles.
- On i circle, draw a Y with the spokes radiating from the dot at 120° angles. On the other circumvolve, draw an inverted Y. These correspond the bonds from C-2.
- Draw lines coming off each circle that bisect the angles of the Y. These represent the bonds from C-3.
You will choose 1 of these templates equally the basis for your project.
Step 2. View the molecule along the C2-C3 centrality with your centre closest to C2.
Your eye will be at the lower left of the image.
The closest bonds make an inverted Y, so you lot will choose the second template for your projection.
Step 3. Place the groups onto your template.
- The groups on C-2 get on the front carbon, The groups on C-3 keep the dorsum carbon.
- The "normal" groups go on the vertical lines: CH₃ in front; CH₂CH₃ behind.
- The wedged groups get on the right hand side: H in front end; CH₃ in back.
- The dashed groups go on the left hand side: Cl in forepart; H in back.
And y'all have your Newman project.
Source: https://socratic.org/organic-chemistry-1/newman-projections/bond-line-notation-to-newman-projection
Posted by: zielinskithencerest1951.blogspot.com


0 Response to "How To Draw Newman Projections From Bond Line"
Post a Comment